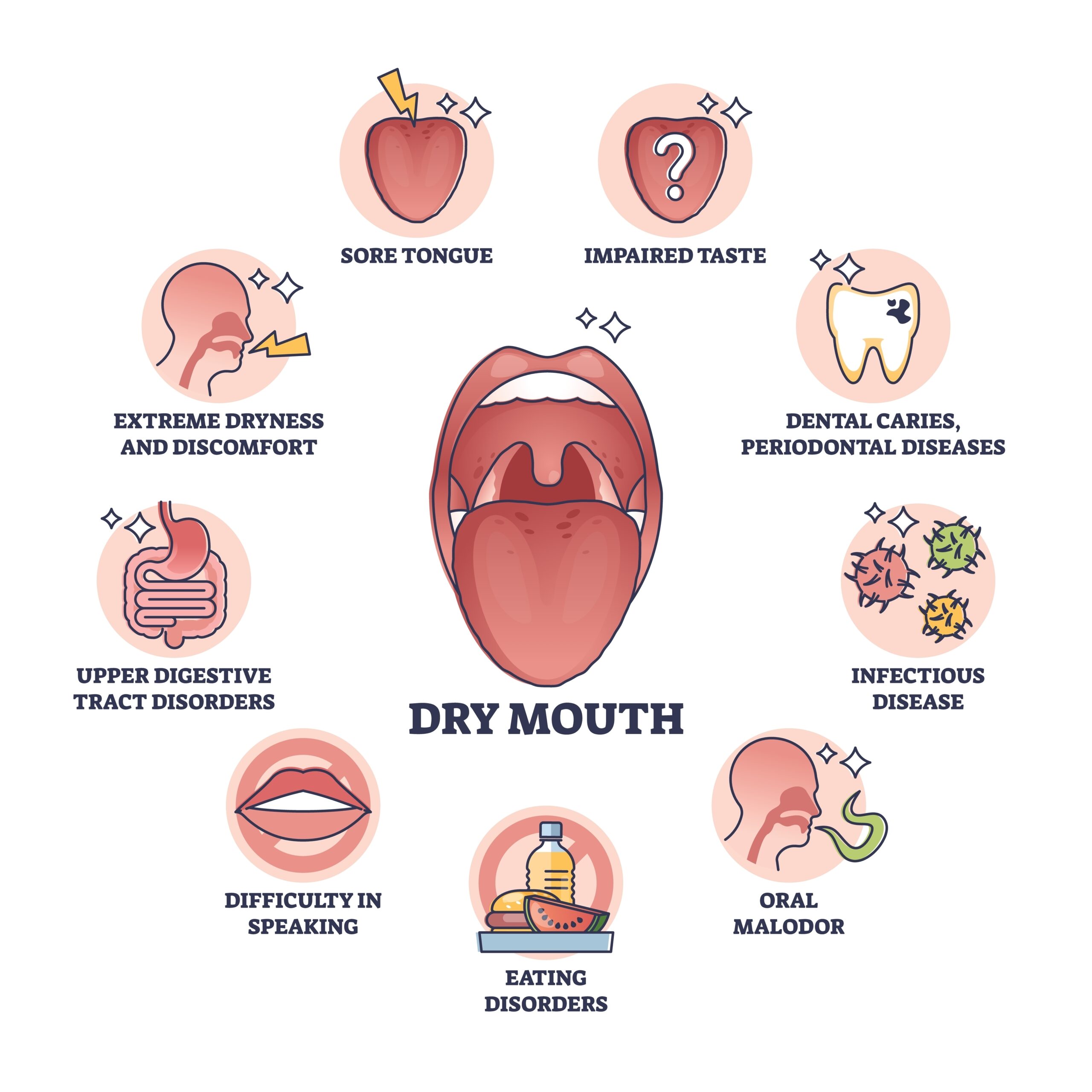
Ozempic, Wegovy, Mounjaro, and Zepbound for Weight Loss in Lupus: What Patients Should Know
Many people with lupus ask me about the newer weight-loss and diabetes medicines such as Ozempic, Wegovy, Mounjaro, Zepbound, and Saxenda. These medicines are often called GLP-1 medicines or GLP-1 receptor agonists.
The big questions are understandable:
Are …