New Research: A Root Cause of Lupus? [Reported July 2024]
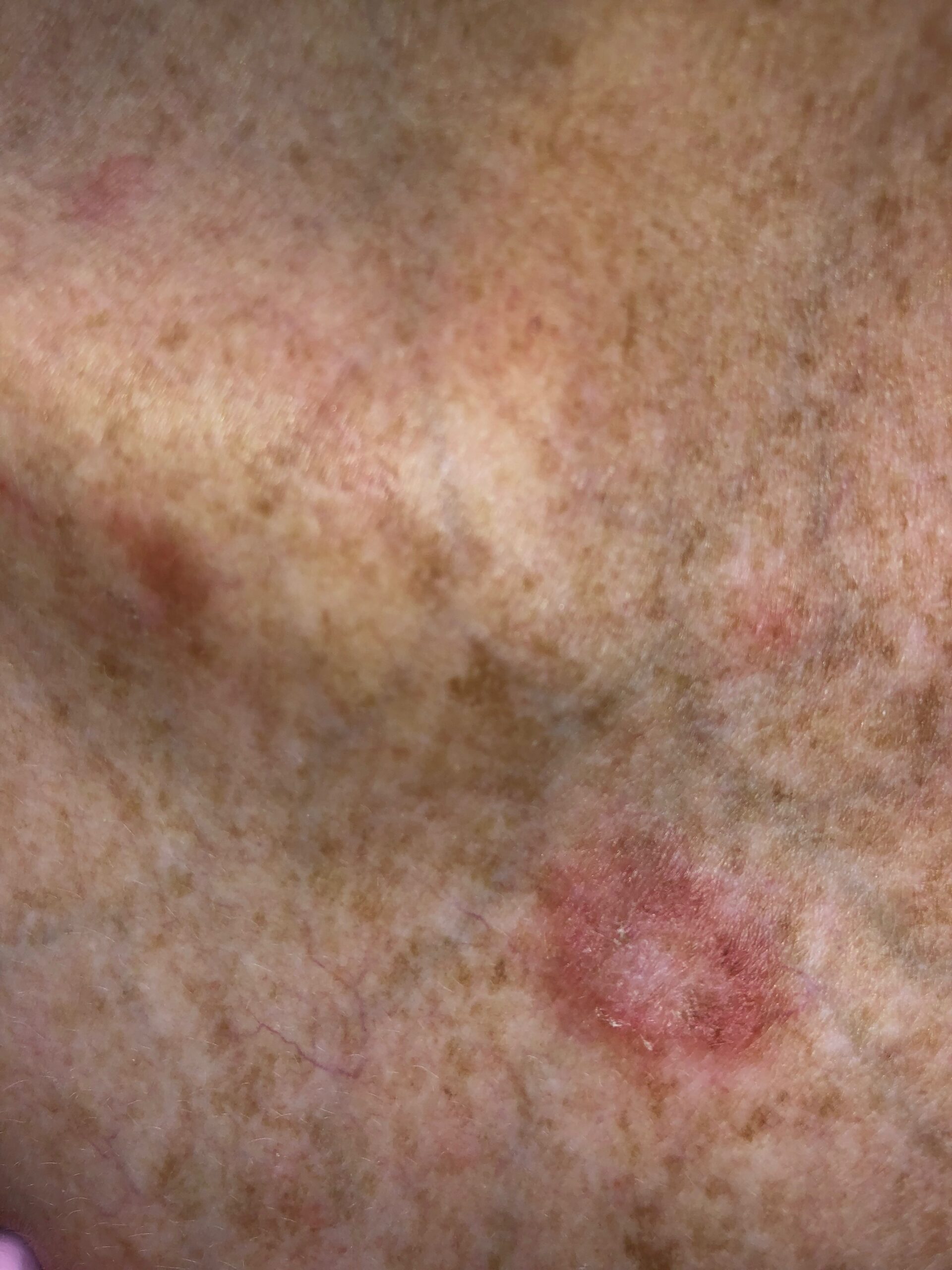
New research claimed they discovered a root cause of lupus. Researchers at Northwestern Medicine and Brigham and Women’s Hospital reported this in July 2024. However, is it really a root cause of lupus? This article discusses the research more closely.…